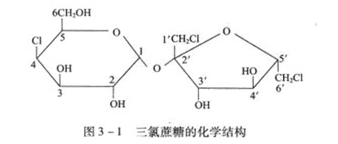
Properties of sucralose
Sucralose is prepared from sucrose by chlorination, and its chemical structure formula is shown in Fig. 3-1.
X-ray diffraction analysis shows that it is an orthogonal crystal with space, each crystal unit contains four three gas sucrose+, and there is an intramolecular hydrogen bond between 0H-2 and 0-3 ', which can prevent the rotation of glycosidic bond between two rings. NMR analysis shows that its hydrogen bond in DMSO is mainly an intramolecular hydrogen bond between OH - 3 'and 0 - 2. Various evidences indicate that there are one or more such hydrogen bonds in the aqueous solution, which plays a great role in the particularly stable property of sucralose in acidic aqueous solution.
Physicochemical properties of sucralose
Generally, it is a white powder product that crushes the large crystalline particles of sucralose into fine particles. Its sweetness is about 600 times that of 5% sucrose solution. The sweetness is pure, and its sweetness is very similar to that of sucrose. There is no aftertaste.

3-2 Solubility of sucralose in water and ethanol
As shown in Figure 3-2, the solubility of sucralose in water is very low, and it is almost insoluble in corn oil. In the multiphase material system with oil or fat using emulsifier, sucralose will enter the aqueous phase, which is similar to sucrose.
The surface tension of sucralose in 20 g and 0.1% aqueous solution is only 71.8 mN/m, which can be ignored due to its small tension to water. If the surface tension in carbonated drinks is large, it will cause many foam, and the surface tension of sucralose is small, which can be well applied to the manufacturing of carbonated drinks, and is suitable for high-speed bottling and canning production lines.
Use Abbe refractometer to measure the refractive index of sucralose, as shown in Ge 3-3. Because its refractive index has a good linear relationship with the concentration, the refractive index method can be used to accurately and quickly determine the concentration of sucralose in aqueous solution or food.
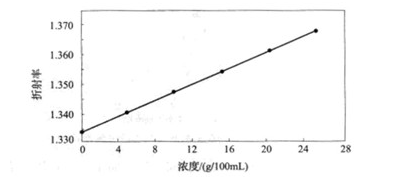
Fig. 3-3 Refraction of Sucralose Solution
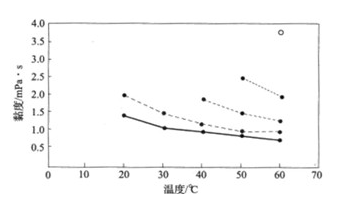
Fig. 3-4 Viscosity of Sucralose Aqueous Solution

